Reactivity trend across periodic table6/19/2023  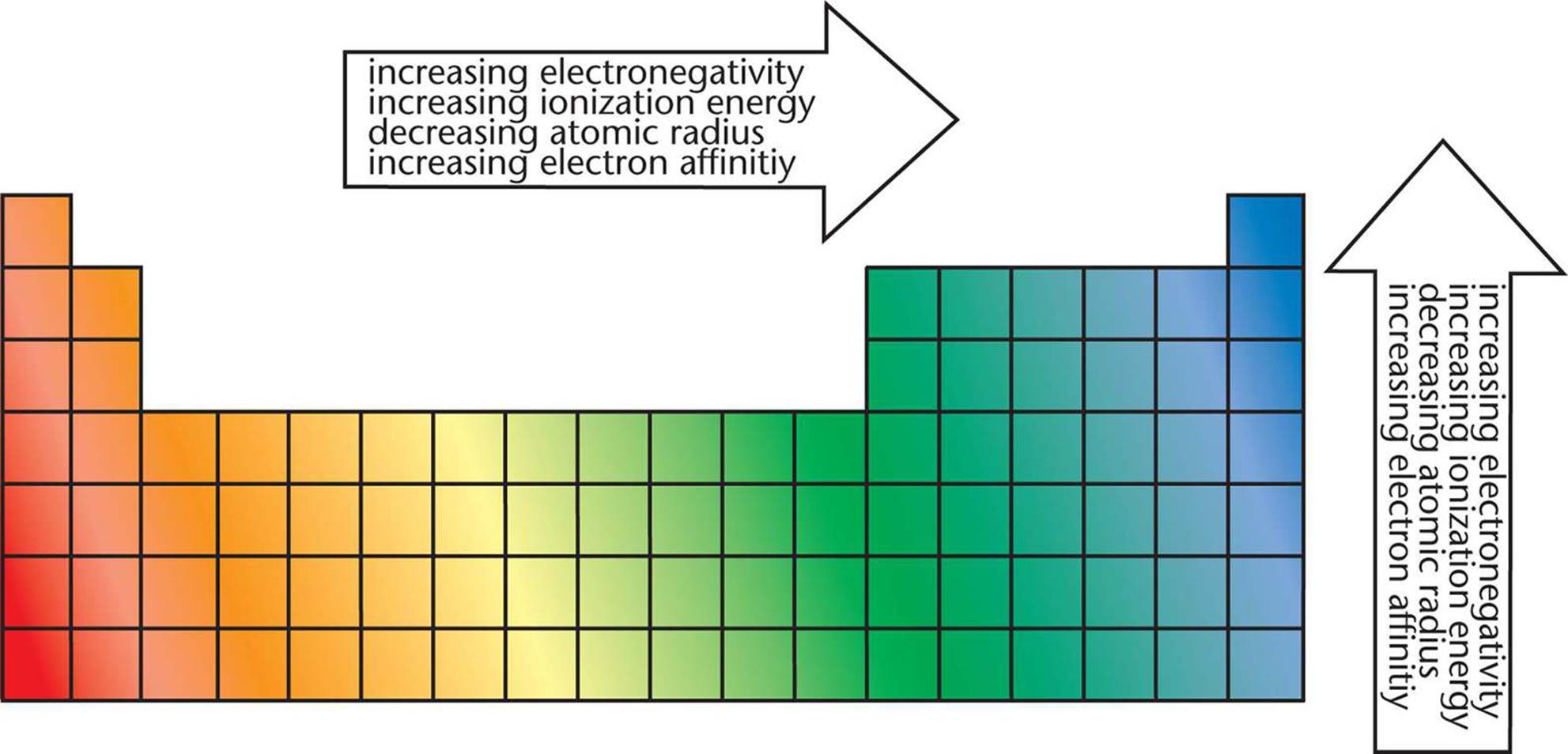
Another trend across Period 3 is each elements reaction with Oxygen to form an Oxide. Periodic Trends, electron configuration period 2, StudySmarter Fig. The overall trend is that reactivity with water decreases across Period 3 from left to right, with Chlorine being the slight exception. These electrons screen or shield the outer electrons from the nuclear charge. You can see in the image below that atomic number increases as you move along the period. This is because of the screening effect of the filled inner electron levels. The greater attraction between the increased number of protons (increased nuclear charge) and electrons, pulls the electrons closer together, hence the smaller size.Īs you move down a group in the periodic table, the covalent radius increases. The covalent radius (a measure of how large individual atoms are) shows different trends if you are moving across a period or down a group.Ī comparison of the relative covalent radii of atoms is shown in the diagram below.Īcross a period from left to right, the covalent radius decreases.Īs you move from left to right across the periodic table, atoms have more electrons in their outer energy level and more protons in their nucleus. The pattern of reactivity of the group 1 elements and the group 7 elements shows the general pattern for the reactivity of other groups.Patterns and trends in the periodic tableĬhemists observe patterns in different properties of elements as they are arranged in the periodic table. For this reason, non-metals are more likely to gain electrons to form a full outer shell. The outer shell of a non-metal is, therefore, more than half full of electrons. Patterns and trends in the periodic table Chemists observe patterns in different properties of elements as they are arranged in the periodic table. These non-metals will have 5, 6 or 7 electrons in the outer shell. Non-metals are mostly found in groups 5, 6 and 7. For this reason, metals are more likely to lose electrons to form a full outer shell. The outer shell of a metal is, therefore, less than half full of electrons. These metals will have 1, 2 or 3 electrons in the outer shell. The periodic table shows that metals are found in groups 1, 2 and 3. Structure of the atoms of metals and non-metals hence, their reactivity decrease across. You can imagine a zig-zag line, starting at B-Al-Si, separating metals from non-metals. metals and non metals have their own trend across the periods, metals easily give up electrons to attain stability. During the course of a period, the atomic and ionic radii of. the non-metal elements are placed on the right-hand side Learning Objectives By the end of this section, you will be able to: Classify elements Make predictions about the periodicity properties of the representative elements We begin this section by examining the behaviors of representative metals in relation to their positions in the periodic table. Trends of Chemical Reactivity in Periodic Table Atomic Radii and Ionic Radii Are Both Defined As Follows.the metal elements are placed on the left-hand side. 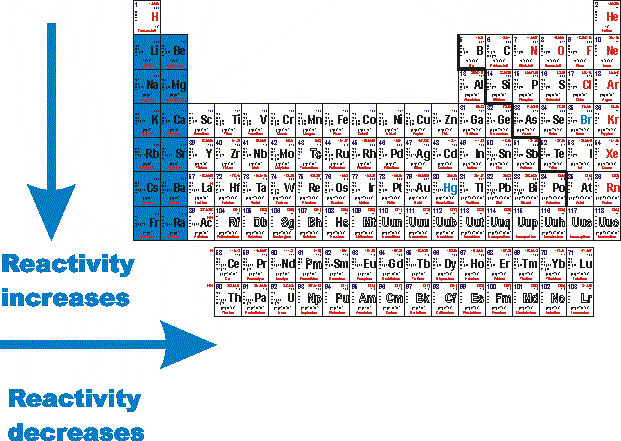
The modern periodic table with some elements left out for simplicity In the modern periodic table, elements are arranged according to their atomic number. Metals and non-metals in the periodic table They have different electronic structure, and this influences the way in which they react. The d orbitals fill with the elements in group 11 therefore, the elements in group 12 qualify as representative elements because the last electron enters an s orbital. The data provided by the reactivity series can be used to predict whether a metal can displace another in a single displacement reaction. The transition elements are elements where the d orbitals (groups 311 on the periodic table) are filling, and the inner transition metals are the elements where the f orbitals are filling. Metals and non-metals are found in different parts of the periodic table. The reactivity series of metals, also known as the activity series, refers to the arrangement of metals in the descending order of their reactivities. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
